Want to know what a geologist would tell you to look for to know if a calcite crystal is authentic or fake?
You can tell if calcite is real when it bubbles in acid, shows a double image when you look through it, can scratch your fingernails, and leaves white streaks. These are all things you can test for at home, but a certified lapidist or jeweler can confirm your findings.
Calcite is very popular in jewelry and spiritual practices, making it a target for dishonest dealers who want to make money. These dealers take clear calcite and dye it or heat treat it to make it look like more expensive or other varieties of crystals, or they make fake calcite from glass or clear resin.
Though fake calcite isn’t as common as fakes of more valuable stones, like diamonds, there has been a rise in the number of fakes on the market in recent years.
In this blog post, I’ll share 12 geologist-approved ways to tell if calcite crystal is real or not, along with the tools professionals use for these tests and what you can use from around your house. I’ve included links to online stores where you can buy these products, if you want to.
No test is foolproof and even the professionals can get it wrong sometimes, so I recommend doing a few different tests before deciding if you’re holding real calcite or not.
By the end of this post, you’ll have 12 geologist-approved ways to tell if calcite is real or fake.
Test if calcite is real with …
A Visual Inspection
Calcite is a bit of a chameleon in the mineral kingdom, coming in quite a few different colors.
Clear calcite often looks milky or clear, much like glass. But don’t be surprised if you find calcite in shades of black, brown, gray, yellow, honey gold, green, blue, lavender, orange, pink, or even red.

Start by taking a close look at the crystal.
In raw form, calcite has a fairly rough surface. When you tilt and turn the crystal under light, the shiny reflections should remind you of the shimmering waters of a clear pond.
A smooth tumbled piece of calcite is also very shiny in the light, with the same lustrous glow as glass or pearls.
Calcite can be transparent, translucent, or opaque.
- Transparent is when you can see through the crystal, almost like looking through glass. Most transparent calcite is clear or a very light milky white.
- Translucent means it lets light through. Clear and milky white calcite varieties are often translucent, though many let some light pass through but not enough to clearly see objects behind the crystal.
- Opaque calcite is not see-through at all and doesn’t let any light through, it simply reflects it. I have a piece of orange calcite that’s opaque – it shines beautifully in the light, but I can’t see through it and it lets no light through either.

Any foreign substances (called impurities or inclusions) in calcite can affect how transparent or translucent the crystal is.
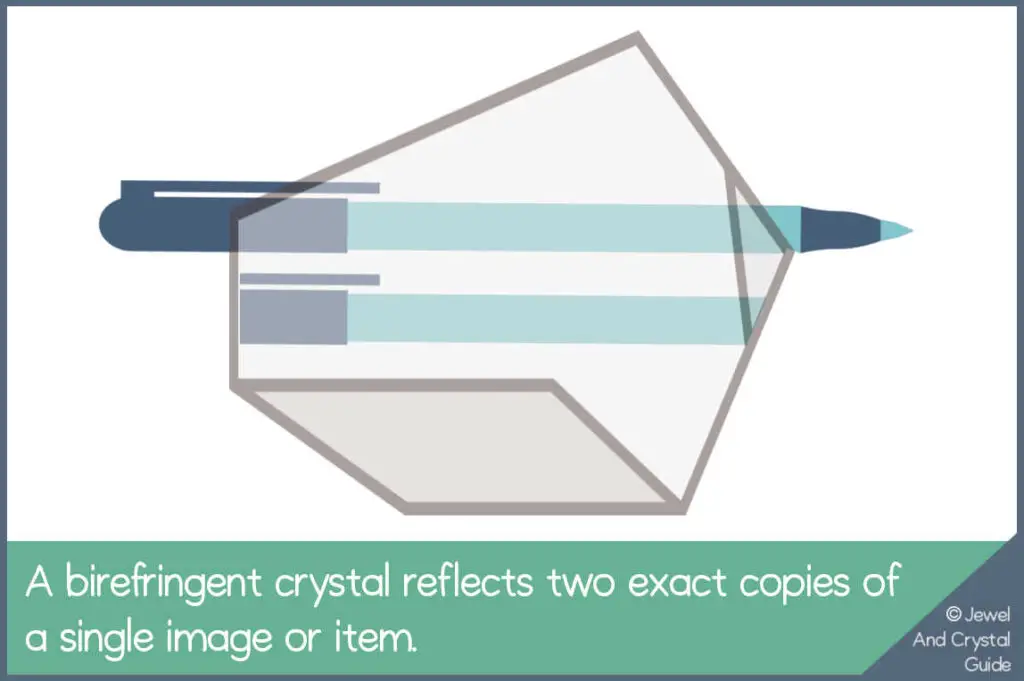
The Double Reflection Test
One of calcite’s most amazing properties is its ability to create a double reflection when looked through, which is called birefringence.
If you have a piece of calcite that’s fairly transparent and translucent, here’s a quick test you can do to tell if it’s real or not:
Put the crystal on top of an object, text, or an image. If you see two copies of the object, text or image through the crystal, it’s birefringence in action and a sign that the crystal is real calcite.
The Acid Test
When real calcite crystal comes into contact with acid, it produces carbon dioxide and makes the liquid acid bubble. This means that you can use acid to test if a crystal is real calcite.
To do the acid test, put one or two drops of an acid solution on to a hidden area of the crystal.
Geologists often use a diluted 10% hydrochloric acid solution for this test. This is a mix of 10% hydrochloric acid and 90% distilled water or another solvent. Anything stronger than this could erode the crystal and cause permanent damage to the area.
If you don’t have any hydrochloric acid, try using pure white vinegar instead. You don’t need to dilute the vinegar for this test. Vinegar contains acetic acid, which is weaker and less reactive than hydrochloric acid, so the reaction might be less bubbly and might take longer to see.
If it’s real, unpolished calcite, it should immediately react and make the acid bubble. This means you have calcite or another mineral that has the same reaction. If there are no bubbles at all, you know it isn’t calcite or the acid isn’t strong enough for a reaction.
Once you have your results, rinse the crystal with water to stop the reaction.
A Scratch Test
The Mohs scale measures how hard minerals are. This scale runs from 1 to 10, with 1 being the softest (e.g. talc and chocolate) and 10 being the hardest (e.g. diamonds).
Items on the Mohs scale are ranked above all the things that are softer than them and below all the things that are harder than them.
Calcite is a 3 on the Mohs scale, which makes it softer than many minerals including copper, steel, fluorite, and quartz. But calcite is harder than and can scratch things that are softer than it, like a fingernail, gypsum, and talc.
Geologists use professional scratch testers, like this set, to determine how hard a mineral is. But if you don’t own such a set, you can make do with things you have around you.
Try scratching the crystal with a copper penny. If the penny scratches or marks the crystal, you have evidence that it’s likely to be calcite. This is because copper is harder than calcite but softer than many other minerals, including quartz (which many people confuse with calcite).
A Scale And A Calculator
Calcite has a specific gravity of 2.6 to 2.7 grams, depending on what’s in the crystal.
You can measure the specific gravity of your crystal for a fairly accurate result. Don’t be scared – I’ll show you how…
To get an object’s specific gravity, we measure how much space it takes when we drop it in water.
Here are the steps to calculate the specific gravity of a piece of your crystal:
- Weigh a container and write down the weight up to two decimal places. Make sure the container is large enough to hold water and the calcite you are testing.
- Add water to the container and take this measurement, again with two decimal places.
- Put the crystal in the water. Now write down this measurement up to two decimal places.
- Subtract the weight of the empty container from the weight of the water-filled container and the water and crystal weight. You now have the true weights of the water and the water and crystal together.
- Divide the true weight of the crystal and water weight by the true weight of the water-only weight. This gives you the crystal’s specific gravity.
If the specific gravity is around 2.6 to 2.7 grams, it falls within the typical range for calcite and there’s a good chance the crystal is calcite, or at least a genuine crystal.
If the specific gravity is significantly out of this range, there might be impurities in the crystal, or it might be imitation glass (which has a specific gravity of around 2.5 grams) or another crystal.
Remember that it’s a good idea to use different methods from this blog post to decide if a piece is calcite or not. These tests are designed to give you a good idea whether the crystal is real or not, so the more tests you do the better your results will be.
A UV Light Test
Calcite can fluoresce, or emit visible light, when exposed to ultraviolet (UV) light, but not all calcite crystals do this. Again, this is just another test you can do to see what results you get, then you can put all the results together to make a better-informed decision.
Here’s how to use a UV light to get more clues on whether or not your calcite is real:
- Get a UV black light from Amazon or a local hardware store.
- Wait for nightfall, then turn off the lights in a room to make it as dark as possible.
- Hold the calcite in your hand or put it on a clean, white surface. Shine the UV light onto the crystal from a few inches away.
- See if the crystal glows under the UV light.
When calcite fluoresces under UV light, it usually glows red or orange-red, but the color depends on what impurities are in the crystal. Some crystals glow green, pink, yellow, blue, or a combination of colors.
So, if you put the crystal under UV light and it fluoresces one of the colors listed above, there’s a chance it’s calcite.
A Heat Test
Metals, like steel, are very good conductors or carriers of heat. If you heat one end of a metal wire, you’ll quickly feel your fingers start burning on the other end of the wire.
Calcite doesn’t carry or hold heat very well because of its basic structure, so the crystal usually feels colder than its surroundings when you touch it.
Put the crystal on your lower lip to see if you can feel a temperature difference. I’ve just tested my own calcite crystal and it definitely feels cool on my lower lip and even in the palm of my hand.
Here’s a simple heat test to tell if your crystal is a good heat conductor:
Find a heat source for this test, like a heater or a pocket warmer. Put the crystal directly on to the heat source, and keep your fingers on the opposite side of the crystal.
Do your fingers start warming up from the crystal?
If it takes a while for your fingers to feel the heat, there’s a chance it’s real calcite. If you feel the heat quickly, it probably isn’t calcite.
Pressure
This test works best if you’re willing to break off a small piece of your crystal, but I’ve included a few tips on how to look for clues of previous breaks in the crystal, to tell if it’s real calcite or not.
When calcite breaks, it splits into perfect rhombohedrons. A rhombohedron is a three-dimensional shape with six sides, and each side is a rhombus.
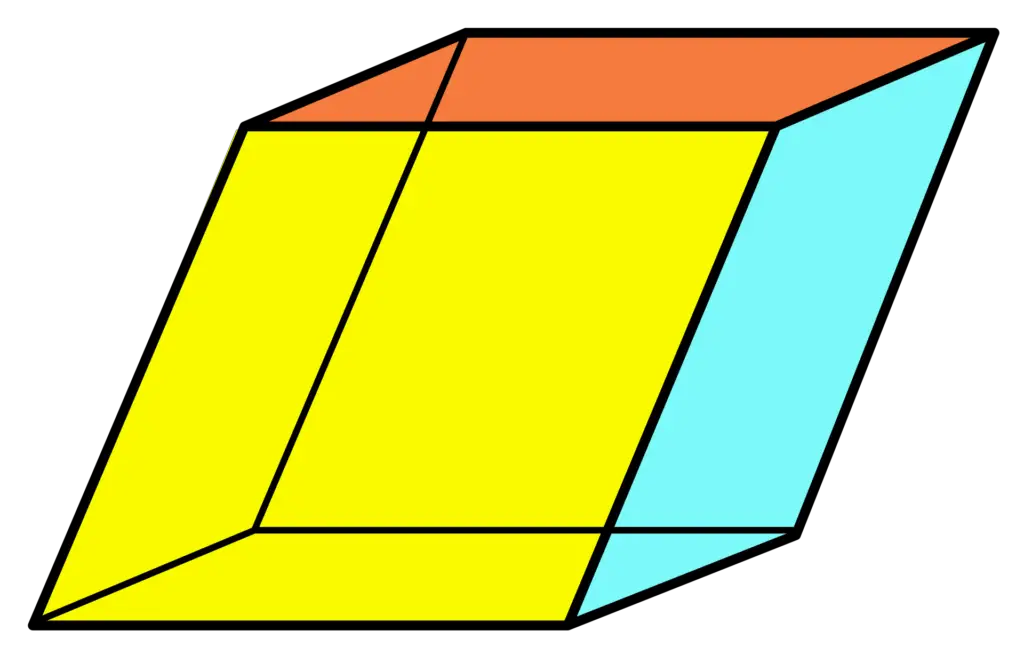
A rhombus is a shape with four sides that are the same length.
Now, I know what you’re thinking – that sounds like a square! Well, a square is a rhombus. It has four sides of equal length that meet at 90-degree angles.
But a rhombus is a shape with four sides of equal length that meet at any angle, not just 90 degrees.
So, a square is always a rhombus, but a rhombus isn’t always a square.
The tendency for a crystal to break in a certain way is called cleavage. We say that calcite has excellent rhombohedral cleavage because it very predictably breaks into rhombohedrons. In fact, this distinct cleavage pattern helps geologists identify calcite accurately.
This happens simply because bonds in calcite’s atoms are weaker along these rhombohedron planes.
When a real calcite crystal breaks, the new smaller shapes have flat sides with mirror-like reflections. Examine your crystal closely and you might see flat, shiny surfaces, which are called cleavage planes.
Next, measure the angles between the planes with a protractor, like this one. You can either measure the angles directly on the crystal, or trace around the crystal and then measure the angles in your drawing.
The obtuse angles (bigger than 90 degrees) between the cleavage planes should be close to 101.5 degrees, while the acute angles (smaller than 90 degrees) should be about 78.5 degrees.
If you want to break off a piece of the crystal, use a pointed tool like a nail or pick to apply increasing pressure on an edge or corner of the calcite until a piece comes off.
If the piece that breaks off is a rhombohedron, there’s a good chance it’s calcite.
Now measure the angles with a protractor if you want to be sure. Again, obtuse angles should be close to 101.5 degrees and acute angles around 78.5 degrees.
A Streak Test
A streak test is done by drawing a line or streak on unglazed porcelain with a crystal. The crystal must be clean and unpolished for this test to work, so you know that other “pollutants” haven’t influenced the results.
The best streak tests are done on freshly broken crystals, where there is no weathering.
You can buy proper streak plates for this test on Amazon, but if you want to use things from around the house try the back of a ceramic tile, the bottom of a porcelain item, or even white cement. These make-do items are not always accurate but it’s worth a try.
A streak test tells us what color the crystal is in powdered form. And you might be surprised to find out that the color of a crystal or mineral isn’t always the same color as its streak!
You could take a red crystal and make white streaks or a silver crystal and make black streaks.
To do a streak test, hold the crystal firmly in your hand.
Choose a pointed edge on the crystal and scrape the point across the streak plate as you hold the plate in place with your other hand. Keep the pressure firm and just run the crystal across the plate once – don’t rub the crystal up and down.
If the streak is white, it’s another sign you’re holding calcite. If the streak is any other color, it is NOT calcite. All calcite makes white streaks, no matter what color the crystal is.
A market comparison
The cost of real calcite depends on the quality, color, and size of the piece, as well as how many people want to buy it and how much calcite is available.
Genuine calcite, especially those with unique formations, are quite valuable.
If the crystal you want to buy or have bought is much cheaper or much more expensive than most other places, there’s a chance it isn’t real.
Here is a table with the average price ranges of calcite per carat for guidance (but remember that these prices change with the market):
| Quality | Price Range (USD) |
| Low-grade calcite | $1 – $5 per carat |
| Medium-grade calcite | $6 – $25 per carat |
| High-grade calcite | $26+ per carat |
A trustworthy seller
If you buy calcite from a seller you know you can trust, and that seller tells you the calcite is real, then it should be. If you aren’t sure and you must know, do your own tests or ask the seller outright.
Some things you can do to make sure the seller can be trusted are:
- Do some research on the seller to look for bad reviews or complaints that other people have made. You can also speak to others or join Facebook groups and ask for recommendations.
- Check the seller’s website to see what information they give and if they look authentic.
- Ask the seller to provide proof on where they got the calcite from.
- Buy calcite jewelry that is certified by the Gemological Institute of America (GIA) or the gem certification body in your country.
Confirmation by a professional
If you must know if a specimen is real calcite or not, and none of the above methods have given you the answer, then ask a professional dealer, lapidist, or appraiser to take a look at the calcite.
A professional has the tools, knowledge, and experience to give you a better answer if a calcite crystal is real or not.
Want to remember this? Save this post on How To Tell If Calcite Is Real to your favorite Pinterest Board below!












